Navigating the 2019 FDA Label Formatting Rules

On May 20, 2016 the FDA announced the final rule regarding the new labeling regulations for packaged food products.
As a food manufacturer, you are probably well aware that the U.S. Food and Drug Administration (FDA) has a lengthy set of rules and requirements that must be followed for nutrition labeling in the United States. These food labeling rules are important in presenting nutrition facts to Americans but can be confusing for those in the food industry. This article will take a look at the basic Nutrition Facts labeling requirements and present you with the best option to go about navigating these requirements.
Background
On May 20, 2016 the FDA announced the final rule regarding the new labeling regulations for packaged food products. The new label’s goal is to better inform consumers and aims to help making healthier food choices easier. Food manufacturers with $10 million or more in annual food sales have a compliance date of 2020 to start using the new label while manufacturers with less than $10 million in annual food sales have until 2021.
General Nutrition Facts Label Requirements
The FDA does not have any specific size requirements for the nutrition label besides the fact that the “Nutrition Facts” heading must be in a type size larger than the rest of the print in the nutrition label. The rest of the nutrition label information must have a minimum type size of 6 point and 8 point. The FDA recommends most information be presents in 8 point Helvetica. This includes nutrition facts information like calories from fat, serving size, added sugars, dietary fiber, total fat, trans fat, saturated fat, etc.
The nutrition facts panel must also be in a black box set off by hairlines and the text up be in all black and printed on a white or other contrasting background. The FDA provides a graphic with the label format enhancements that the FDA prefers to be used. You can see this graphic below:
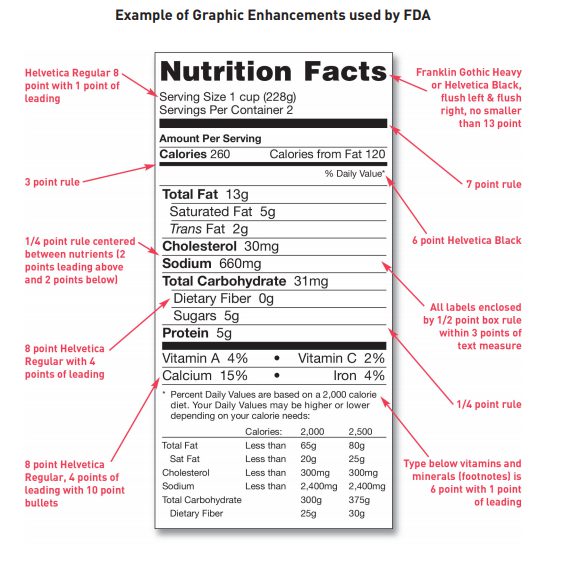
Nutrition Facts Label Unable to Fit
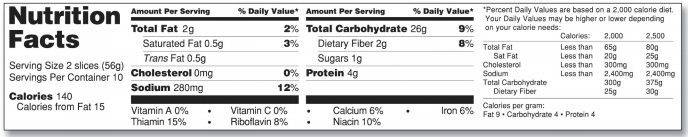
If the packaging you are trying to place the nutrition facts label on doesn’t have enough room, the FDA allows a “side-by-side” labeling format. The FDA specifically lists this packaging as those with “more than 40 square inches”. The FDA allows these Nutrition Facts labels to place the bottom part of the label (the information following the vitamins and minerals) to the right and separated with a line. The FDA provides the following example nutrition facts label:

If the packaging does not contain continuous vertical space (around 3 inches) to use a “side-by-side” label, the FDA allows a tabular (horizontal) format to be used. Here is the example that the FDA provides:
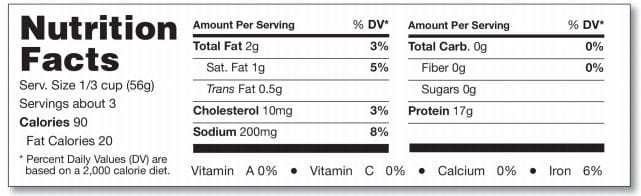
For small and intermediate-sized packages (those with less than 40 sq. in or less total surface area available for labeling) that can’t accommodate nutrition information in columns on any label panel, the FDA permits a linear (string) format. Please see the following example:

Food packages that have less than 40 sq. in or less available for labeling may omit the footnote and use a tabular display format as long as an asterisk is placed at the bottom of the label that states “Percent Daily Values are based on a 2,000 calorie diet”. The FDA provides the following example:
Navigating the Requirements
Trying to follow these confusing FDA requirements on your own can be quite frustrating and possibly lead to compliance errors or other issues. Using an FDA-compliant labeling software like LabelCalc makes following these FDA requirements easy. All formats are built into the software and ensures that the correct Nutrition Facts label is used for the corresponding package size. LabelCalc’s software allows you to enter in your recipes and you will be provided with instant nutrition analysis with FDA-compliant labels.
LabelCalc is an industry-leading recipe analysis tool used by food manufactures, global retail stores and food entrepreneurs. To get started, contact our sales team today.
